|
Surfactants (surface active agents)
are used in the emulsification process. A surfactant can be considered
a "confused" molecule having both a hydrophobic (water-hating) and
hydrophilic (water-loving) part. Surfactants are also referred to as
wetting agents and foamers. It is said that they "make water wetter".
The surfactant we used in all of the following experiments is dish
soap, but other examples of surfactants include detergents, hand soaps,
etc. Surfactants lower the surface tension of the water, thus making it
easier for cleaners to lift dirt and grease off of dirty dishes and
clothes and help to keep them suspended in the dirty water to keep them
from being redeposited on the clothes and dishes. In an emulsion, they
temporarily stabilize the oil and water mixture. As anyone who uses
Italian dressing knows, the oil and water do not mix unless shaken
vigorously in the bottle. They separate almost immediately afterward.
Although you can create an emulsion without a surfactant, surfactants
lengthen the amount of time it takes to separate these two phases
(sometimes it could take years to separate the oil and water in an
emulsion).
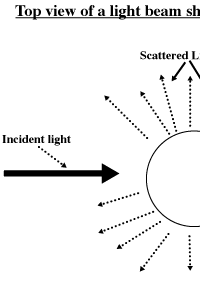
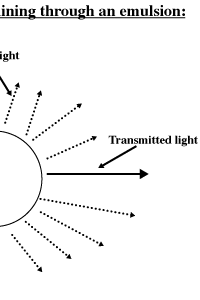
The Tyndall Effect:
The Tyndall
Effect is defined as the scattering of light by the particles of a
mixture. Many of us have seen this already as the dust in the room
scatters some of the light from a movie or slide projector. The fact
that most clouds and fog appear white is also the result of the Tyndall
Effect.
Emulsions are
mixtures that scatter light. Solutions (which can be defined as
homogenous mixtures) do not scatter light. A light beam passing through
a solution can be seen only along the direction of original incidence
(the darker line in the illustration below). The emulsion that we make
in lesson #4 turns white as the oil and water are mixed in the presence
of a surfactant. A color change can represent a chemical change (as
explained later). However, for this lesson, the color change is due to
the Tyndall Effect (See below). The observer sees the emulsion as white
due to the scattering of light in all directions by the particles in
the emulsion. If you were to look into a dilute water-emulsion, you
would be able to see the light beam passing through the liquid and
being scattered in all directions around the container.
Physical vs. Chemical Changes:
One of the topics
brought up by our unit is the difference between chemical and physical
changes. So, a little compare and contrast is helpful at this point.
Chemical changes
occur when one substance becomes a different substance (with different
properties). Evidence of a chemical change can be: energy being
released or absorbed, a color change, and the formation of a
precipitate or formation of a gas. Another name for a chemical change
is a chemical reaction. The emphasis here is that a different substance
is formed. Burning of wood is a chemical change.
Changes in size,
shape, color or state are generally physical changes. The substance is
still the same, although its appearance is altered (like crumpling a
piece of paper). Making a mixture is an example of a physical change.
In a mixture the original materials are mixed together but not
chemically altered.
During the course
of this unit, you and your students will be exposed to making "white"
emulsions. In the case of lesson #4, it is a physical change since
nothing new is created. The oil and water are just more thoroughly
incorporated within each other with the help of the surfactant. This
causes globs to form that scatter light and make the emulsion appear
white. In lesson #5, you and your students will be combining two
different chemicals to make a new one. Stearic Acid + Sodium
Bicarbonate = Sodium Stearate. Stearic acid is obviously and acid,
sodium bicarbonate is a base. When these chemicals are combined in the
correct proportion neutralization occurs. Neutralization is an example
of a chemical change. The solution properties of sodium stearate are
different than the constituent chemicals that you began with.
Suggested sequence of lessons for "Emulsions"
LESSON 1: Testing household products
In this lesson,
students are exposed to the idea of an emulsion, and then use the
scientific method to distinguish between o/w and w/o emulsions. An
extension activity on label reading is also included.
LESSON 2: Surfactants and water tension
In this lesson,
students learn about surface tension by making paper clips float on
water and then adding detergent to the water and watching them sink.
They then will test the water tension of different types of water
(soapy, tap, and salty) and observe their behavior.
LESSON 3: Observing emulsions
In this lesson,
students will make simple examples of a w/o and o/w emulsions. They
will compare and contrast their observations of the difference in
behavior of the two types as well as observing the behavior of
surfactants in the mixture. Observing the density differences between
the oil and water phases is emphasized.
LESSON 4: Making the end emulsion
Students will use
the knowledge gained during lessons 1-3 to make a simple emulsion in
the classroom. They will observe its stability over the course of a
night to see that a physical change has occurred.
LESSON 5: Saponification — Using a chemical change to create a hand cream.
In this lesson,
students will take the emulsification process one step further. By
using simple chemicals (stearic acid and baking soda) students can
observe a chemical change as represented by a change in pH in making a
cosmetic.
Supplies:
Lesson 1 to 4 can be accomplished with simple household supplies. In
lesson 5 you will need stearic acid which can be ordered from most
scientific companies. We have included the name of just one company.
Nasco - 901 Janesville Ave. Fort Atkinson, Wisconsin 53538-0901
Phone - 920-563-2446 Fax - 920-563-8296
| Mineral oil - |
SA 07851 M |
500 ml |
$5.40 |
| Stearic acid - |
SB 17050 M |
100g |
$8.05 |
| |
SA 09770 M |
500g |
$11.35 |
These materials will be sufficient to do these activities for several years.
LESSON 1: Testing household products
Grouping:
Students will be in groups of 2-4
Materials:
At least 10
different household emulsions (sunscreen, hand cream, salad dressing,
etc.). (Avoid cleansers, caustic substances and bleaching substances)
At least 10 test tubes w/lids or small bottles with lids per group
10 beakers with medicine droppers or pipettes (one per emulsion to be tested)
Tap water to fill test tubes/bottles
Safety glasses
Procedure:
- Number beakers then fill beakers 1/3 of the way full with each emulsion.
- Get students into groups.
- Distribute 10 test tubes w/lids (or bottles) to each student group.
- Fill containers 3/4 full with water.
- Give one numbered beaker to each group with pipette/medicine dropper.
- Have students drop one drop of emulsion into test tube/bottle of water.
- Have students put lids tightly on containers and shake vigorously.
- Observe water in test tube/bottle.
- If water is cloudy/milky then have the students record that emulsion as o/w.
- If water is
relatively clear and the emulsion appears as round droplets dispersed
in the water, then have the students record that emulsion as w/o.
- Have students pass beakers to the next lab group and repeat the process until all lab groups have tested each emulsion.
- Dispose of
emulsion-water mixture by discarding them down sinks and have students
wash their hands thoroughly with soap and water.
Lesson 1 Data Table:
| Emulsions # - |
Observations |
Emulsion Type |
| 1 |
|
|
| 2 |
|
|
| 3 |
|
|
| 4 |
|
|
| 5 |
|
|
| 6 |
|
|
| 7 |
|
|
| 8 |
|
|
| 9 |
|
|
| 10 |
|
|
Extension Activity:
- Number the original containers to correspond with the beaker numbers.
- Pass around the original containers and have the students note where water appears in the list of contents.
- Have students record the location of water in the list of ingredients as being one of the first or one of the last ingredients.
- Pass around
common household emulsions and have students determine if they are
water continuous or oil continuous based on reading the list of
contents and the relationship they discovered in step 4.
- Review data with students.
Lesson 1 Data Table:
| Product |
Location of Water |
Type of Emulsion |
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
Observation:
- After reviewing the data, do you observe a relationship between emulsion type and where water appears on the ingredient list?
- What do you
think the criteria is for listing materials on a label? (Alphabetical
order, amount of ingredients cost on ingredients)
Lesson 2: Surface tension - Part 1
Grouping:
Students will be in groups of 2-4
Materials:
Small paper cups
New paper clips
Dish detergent
Water
Procedure:
- Distribute one cup and 5 to 6 paper clips to each group.
- Have students fill cups 3/4 full with water.
- Have students bend paper clips as shown in diagram 1.
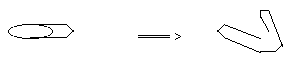
- Have students carefully place paper clips on the surface of water and release the paper clip.
(Students should attempt this until at least one stays on top of the water.)
- Have students carefully place a drop of dish detergent into their cup, but not actually touching the clips.
- Have students record their observations below.
- Students should then attempt to put another clip on the water and observe what happens.
Observation:
- Describe what happens when you place the dish detergent into the water?
- Were you successful in floating a paper clip on the water after the detergent was added to the water?
Surface Tension Part 2
Materials:
One clean penny and quarter per group
Soapy water (dish soap and water)
Salt water
Medicine dropper (pipettes) or micropipetes
Paper towels
Rubber stoppers
Tap water
Calculators
Hypothesis:
How many drops of tap water do you think you can fit on a penny and quarter before it spills?
Penny ____________Quarter ____________
How many drops of soapy water do you think you can fit on the penny and quarter?
Penny ____________Quarter ____________
How many drops of salt water do you think you can fit on the penny and the quarter?
Penny ____________Quarter ____________
Procedure:
- Put rubber stopper on top of paper towel and then place your penny on the rubber stopper head side up.
- Carefully place drops of tap water on the penny, counting as you go. (Be careful not to allow the dropper to touch the water.)
- Record the number of drops your penny held before the water overflowed.
- Repeat steps one to three using tap water and the quarter. Record on data table.
- Repeat step one to four for soapy water and then for salt water. Record on data tables on following page.
Lesson 2 Data Table:
| Tap Water |
| COIN |
Trial 1 |
Trial 2 |
Trial 3 |
Average |
| Penny |
|
|
|
|
| Quarter |
|
|
|
|
| Soapy Water |
| COIN |
Trial 1 |
Trial 2 |
Trial 3 |
Average |
| Penny |
|
|
|
|
| Quarter |
|
|
|
|
| Salt Water |
| COIN |
Trial 1 |
Trial 2 |
Trial 3 |
Average |
| Penny |
|
|
|
|
| Quarter |
|
|
|
|
Conclusion:
- Did you have the same number of drops for each trial for each type of water on each penny?
- If there was a difference, why do you think this happened?
- Did the type of water seem to influence the number of drops of water you could place on the coins?
- What type of water enabled you to place the most amount of water on each coin?
- Which type of water enabled you to place the least amount of water on each coin?
- Based on the paper clip experiment can you explained why this happened?
Lesson 3: Observing Emulsions
Grouping:
Students will be in groups of 2-4
Materials:
Dawn dish soap
Glass or clear plastic bottles (4 per group)
Mineral oil
Water
Stopwatches (1 per group) or class clock
(Optional) food coloring
Markers or grease pencils
Procedure:
- Fill one small
container with mineral oil and one other one with water and distribute
to each group (adding food coloring to the water beforehand makes it
easier for the students to discern which is the oil layer and which is
the water layer!).
- Distribute 2 empty containers of the same size to each group as well.
- Have the student label the empty containers w/o and o/w
- Have the students pour about 3/4 of the oil into the w/o container and the rest into the o/w empty container.
- Add a drop or two of food coloring to the water (optional)
- Have the students then pour water into the containers with the oil in them until they are nearly full, but not completely full.
- Have the students observe what the two containers look like and draw below:
| |
 |
 |
| w/o |
o/w |
| |
- Answer the following questions:
| |
| 1. Where is the oil layer in each container? |
| |
|
| |
| 2. Where is the water layer in each container? |
| |
|
| |
| 3. Are they mixing? Why/Why not? |
| |
|
| |
|
| |
- Have students shake each of the containers vigorously for about one minute.
- Have students observe what is happening in each of the containers and draw below:
| |
 |
 |
| w/o |
o/w |
| |
- Have the students time how long it takes for each to the separation to re-establish itself. Write the time:
- Describe the "bubbles" that you see in the container.
| Are they the same size in each container?_____________________ |
| Is there the same amount of "bubbles" in each container? |
| |
|
| |
- Have the student
add a squirt of dawn dish soap to each container (enough to cover
bottom of container). Ask them to observe what happens.
| Where did the dish soap go? |
| |
|
| |
| Is it moving? |
| |
|
| |
| Where is it going? |
| |
|
| |
- Have them draw what they are observing below:
| |
 |
 |
| w/o |
o/w |
| |
- Dispose of
emulsion-water mixtures by discarding them down sinks and have students
wash their hands thoroughly with soap and water.
Explanation: How to get the "best" emulsion:
When we set out
to make an emulsion, we didn't have any idea which proportion of
mineral oil to water to soap would work the best. What follows is a
description of how we used the scientific method to arrive at the
proportions for lesson #4.
| % oil |
% water |
amount of Dawn (ml) per 200 ml of emulsion |
| 95 |
5 |
2 |
| 90 |
10 |
2 |
| 85 |
15 |
2 |
| 80 |
20 |
2 |
| 75 |
25 |
2 |
| 70 |
30 |
2 |
| 65 |
35 |
2 |
| 60 |
40 |
2 |
| 55 |
45 |
2 |
| 50 |
50 |
2 |
After observing the separation of these emulsions (which was not desirable), we decided to use 4 ml
of Dawn in the same proportions as above. The overall results were much
better (see following graphs). We mixed these batches in relatively
large containers and used manual eggbeaters to help in the mixing. We
realized that this created an excess amount of foam as well as
presented extra equipment for users of this activity, so we decided to
test the "best" of the above proportions using manual shaking for one
minute. In this case, no eggbeaters would be necessary. The results
were not as good as with the eggbeaters.
We then decided
to try adding much more soap (as a % of the total volume) and continue
shaking with the "best" of the above proportions. The results were very
encouraging. The shaken emulsions with the increased soap were much
more stable for a longer period of time than what we had done
previously. We also decided to try Dawn Power Plus, and in the end, we
feel that that produced the most stable emulsion. It is that formula
which we used in lesson 4.
So, why go to all
this trouble? We have found that different oils and different soaps
perform differently. Olive oil can be used instead of mineral oil, but
different proportions might be necessary to achieve the "best"
emulsion. Same thing for the different soaps. You could have your
students test the materials that you have as part of the scientific
method. Each group could be given one or two proportions to test with
the same soap and oil. You could try changing oils and comparing the
results. You could try changing soaps and comparing results. These
extension activities could definitely enhance the scientific method in
the context of our emulsion lesson.
Worksheets:
Worksheets for Lesson 3 are available for download in PDF format:
Lesson 4: Making the End Emulsion
Grouping:
Students will be in groups of 2-4
Materials:
Materials per group:
Mineral oil (approx. 45 ml)
Water (approx. 5ml)
Power Plus Dawn (approx. 5 ml)
Clear glass or plastic bottles (2) with lid
Graduated Cylinders (2)
Graduated pipettes
Procedure:
- Have students
measure out the amount of the above materials from a centralized
location using graduated cylinders and return to their desks (for ease
of mixing, you could have the student getting the 5 ml of water also
get the 5 ml of Dawn in the same graduated cylinder-this will allow the
Dawn to come out of the graduated cylinder to be mixed much more easily
than if you just had the Dawn)
- Have students pour the Dawn and water into the bottle with the oil.
- Secure bottle cap tightly on bottle.
- Have students shake vigorously for about 1 minute.
- Observe what happens and record below.
- Let emulsion stand overnight and have the students observe their emulsion the next day and record below.
Observations:
Describe what you observed below (use color, consistency, etc.)
- Oil:
- Water:
- Emulsion (right after mixing):
- Emulsion (next day):
Enrichment:
We feel that
students will have an excellent opportunity to use the scientific
method if you allow them to determine the "best" proportions and/or the
best surfactant for their emulsions. This can be done by dividing the
class into 6 groups. Have each group test a different surfactant.
(Dawn, Joy, Palmolive) and different proportions of oil and water.
After allowing their emulsions to sit for 24 hours, students can then
report their findings to the class.
Lesson 5: Observing Chemical Change in Making a Hand Cream Emulsion:
Grouping:
Students will be in groups of 4-5
Materials (per group):
Glass bottle with lid
2 small beakers
2 graduated cylinders
Mineral oil (or baby oil)
Stearic acid
Baking soda
Water
Hot plate
pH paper
Triple beam balance
Weighing paper
Pot holders/oven mittens
Safety goggles
Safety Concerns:
When using hot
plates, be sure to make students aware that they should not touch the
hot plates (even after they are turned off) because they are hot. They
also do not need to heat the solutions very much (the students are not
trying to boil the solutions (50 degrees C is equivalent to 122 degrees
F, which is warm, but not overly hot)). During the whole lab, have
students keep safety goggles on for eye protection. When transferring
the warm solutions to the larger bottle, make sure that they use
something like potholders or oven mittens to hold the beakers (so that
they don't burn their fingers). In the same way, if the bottle is too
hot to handle for shaking, allow it to cool without the lid on it for
several minutes until the bottle is cool enough to handle before
shaking!!!
Procedure:
- Measure out 12.5 ml of mineral oil.
- Weigh out 0.75 g of stearic acid.
- Using low heat, have students dissolve the stearic acid in the mineral oil (the solution will turn clear when stearic acid dissolves).
- Get pH of solution with pH paper and record.
- Obtain 37.5 ml of water.
- Weigh out 0.5 g of baking soda
- Slowly heat the water (under low heat) and dissolve the baking soda.
- Get pH of solution with pH paper and record.
- When both solutions are dissolved, pour both into jar with lid using potholders/oven mittens/tongs/test tube clamps!!
- If jar is too
hot to handle with bare hands, allow to cool for several minutes before
putting lid on. Jar should be warm to the touch (NOT HOT) when you begin shaking.
- Close jar lid tightly and shake vigorously for 1 minute.
- Get pH of solution with pH paper and record.
- Allow to sit overnight.
- (optional) If
students want, have them take a small amount of cream with a wood
splint or similar tool and put a small amount on back of hand and
spread with fingers.
- (optional) Have students record their observations of the cream as it was applied to their skin.
Observations:
pH of stearic acid and oil ____________.
pH of baking soda and water ___________.
pH of cream ___________.
(optional)
observations of hand cream on the skin:
This experiment is courtesy of 
|
|



